The pathogenesis of rosacea is multifactorial: one component is believed to be infestation with microorganisms, the skin mites Demodex folliculorum and Demodex brevis. A compound, ivermectin 1% cream, which is currently under review by the US Food and Drug Administration (FDA), is the first agent to target an underlying cause of rosacea, infestation with the Demodex skin mites. If approved by the FDA, this new compound will be the first medication to address a key component of papulopustular rosacea.
In a recent interview, Zoe D. Draelos, MD, Consulting Professor of Dermatology at Duke University School of Medicine, High Point, NC, and one of the senior investigators of ivermectin 1% cream, discussed how it works, and what it is expected to contribute in the management of patients with rosacea.
Q: How does Demodex affect the skin?
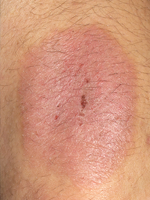
Dr Draelos: Rosacea is a condition in which a mite, Demodex, lives on the face of the patient. This skin mite causes challenges to patients with rosacea. Their faces turn red on the nose and cheeks, and they get small inflammatory papules and pustules that are itchy, uncomfortable, and unsightly. These facial lesions can sometimes progress to cause overgrowth and enlargement of the nose, a condition called rhinophyma.
This facial erythema in rosacea is not just flushing, especially in the more mature patient. In childhood, the face turns red with exercise, but in adults, the erythema remains after a triggering insult, such as Demodex infestation, heat, red wine, certain foods, and so forth. Patients can have erythema, burning, and stinging that can last for days.
Q: How does ivermectin 1% cream address this component of rosacea?
Dr Draelos: What ivermectin does is it reduces inflammation and kills the Demodex. It has been used as an oral drug for many years in veterinary medicine. This is the first topical formulation of ivermectin that has tested safe for human use and that has proved effective in patients with rosacea. It is clearly more effective than any of the other topical agents we use for patients with rosacea, and it is more effective than current agents in a unique way.
Q: In the past, the role of Demodex in rosacea was debated. Has its contribution to rosacea become more established?
Dr Draelos: There has always been some degree of professional controversy about the role of Demodex. Within the gut of Demodex lives a type of bacteria. Some experts think it is the bacteria that create the immune response in the skin, and some think it is the Demodex itself. The bottom line is, this does not matter. Ivermectin kills the Demodex, and when it dies, so do the bacteria.
The recent efficacy seen in phase 3 clinical trials of ivermectin 1% cream lends credence to the theory that I have long held—that Demodex are highly operative in inflammatory rosacea.
Q: What about the safety of ivermectin?
Dr Draelos: As an antiparasitic compound, ivermectin works by acting as an agonist (namely, activating) of the GABA neurotransmitter in the nerve cells of Demodex. I am sometimes asked whether this may affect nerves in humans, but humans do not have GABA receptors. Ivermectin is not neurotoxic for humans, only for the parasite. It will not cause nerve issues in humans. Its safety profile is excellent.
Q: Which patients with rosacea would be appropriate for treatment with ivermectin 1% cream?
Dr Draelos: The FDA indication for this medication is expected to be for patients with erythema, papules or pustules, and an acne component to their rosacea. Skin mites may not be the cause of rosacea for every patient, but it is the underlying cause of rosacea in patients with an inflammatory or acne component, which encompasses approximately 50% of the patients I see in my clinical practice.
Q: Are there advantages to ivermectin, other than its ability to clear the skin?
Dr Draelos: Yes. Rosacea is a common chronic condition that can require continued treatment. With ivermectin, there appears to be a sustained effect, even once the drug is discontinued. There is often a disease-free interval during which the patient’s skin can remain clear and he or she can forego treatment.
These remissions lower the treatment burden of rosacea. This is important not only to the patient, but also to the payer community, because it decreases the cost of the medicine across the lifetime of the patient.
In the phase 3 clinical trials, in which patients received ivermectin 1% cream or vehicle (ie, placebo) cream for 12 weeks, we discontinued treatment when the patient’s skin cleared and restarted the treatment when inflammatory papules and pustules reappeared. We saw from those data that there are longer disease-free intervals with topical ivermectin treatment.
We saw quick efficacy and a sustained effect, and this drug stands out from other treatments thanks to these attributes. Those disease-free intervals are the key to getting better control of rosacea as well as to lowering the cost of treatment.